
The multifaceted functions of PLKs raise the question of how PLKs are dynamically targeted to distinct structures during meiotic progression. Similarly, mammalian PLK-1 localizes along the SC and functions in SC disassembly ( Jordan et al., 2012). PLK-2 subsequently relocates to the SC in pachytene and controls DSB formation, SC disassembly, and meiotic chromosome segregation ( Harper et al., 2011 Nadarajan et al., 2017 Sato-Carlton et al., 2018). In Caenorhabditis elegans, a meiosis-specific PLK-2 is localized to special chromosomal regions known as pairing centers and promotes homologue pairing and synapsis ( Harper et al., 2011 Labella et al., 2011). It also controls the monoorientation of sister kinetochores ( Galander et al., 2019) and stepwise loss of cohesin during meiosis ( Attner et al., 2013 Brar et al., 2006 Katis et al., 2010). The budding yeast Cdc5 is central to crossover recombination and SC disassembly in late pachytene ( Argunhan et al., 2017 Clyne et al., 2003 Sourirajan and Lichten, 2008). Polo-like kinases (PLKs) play widely conserved but diverse roles in regulating the complex chromosome behavior during meiosis. These rearrangements of chromosomes then enable the separation of homologues in meiosis I and later sister chromatids in meiosis II. During meiosis I, sister kinetochores are attached by microtubules from the same spindle pole (monoorientation), and the cohesion at centromeres is protected from cleavage ( Petronczki et al., 2003). A subset of these breaks is repaired to form crossovers, which, together with sister chromatid cohesion, physically link each pair of homologues. 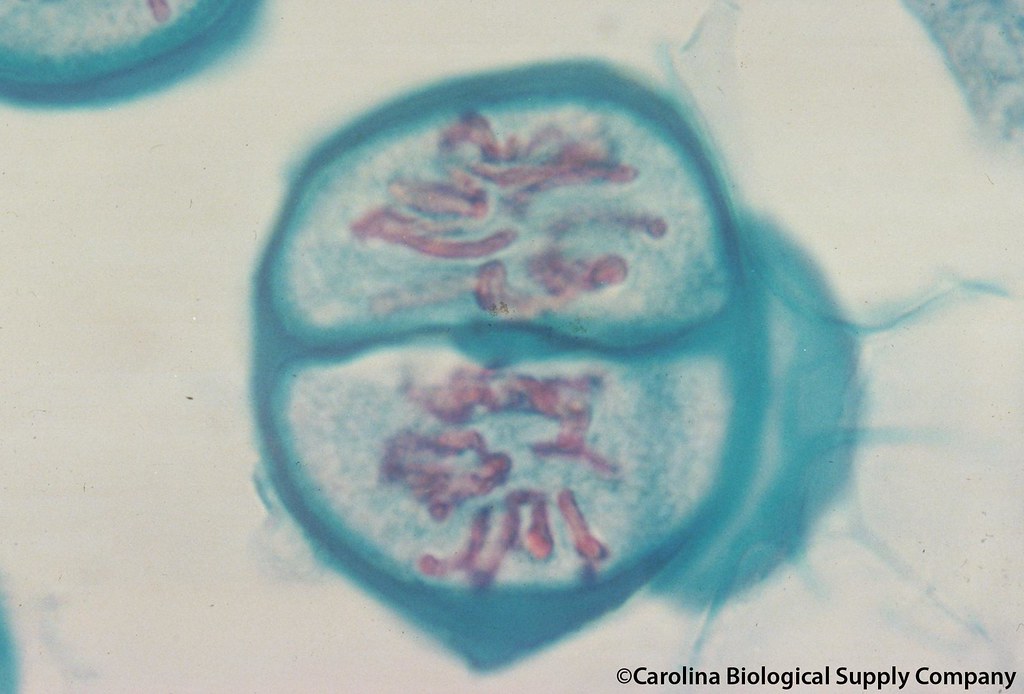
Concomitantly, meiotic recombination initiates with the generation of programmed DNA double-strand breaks (DSBs de Massy, 2013). 
Unique to meiosis is its protracted prophase I, in which chromosomes pair and synapse with their homologues through the formation of a zipper-like protein structure called the synaptonemal complex (SC Bhalla and Dernburg, 2008 Page and Hawley, 2004). The production of haploid gametes during meiosis is achieved by a single round of DNA replication followed by two consecutive nuclear divisions. Thus, the integration of chromosome-autonomous signaling and a nucleus-wide crossover-counting mechanism partitions holocentric chromosomes relative to the crossover site, which ultimately defines the pattern of chromosome segregation during meiosis I. PLK-2 is targeted to crossover sites primed by CDK-1 and spreads along the SC by reinforcing SYP-1 phosphorylation on one side of each crossover only when threshold levels of crossovers are generated. However, PLK-2 docking to the SC is prevented by the nucleoplasmic HAL-2/3 complex until crossover designation, which constrains PLK-2 to special chromosomal regions known as pairing centers to ensure proper homologue pairing and synapsis. SYP-1 phosphorylation by CDK-1 occurs just before meiotic onset. Here, we demonstrate that the cyclin-dependent kinase CDK-1 primes the recruitment of PLK-2 to the synaptonemal complex (SC) through phosphorylation of SYP-1 in C. However, how PLKs are targeted to distinct subcellular localizations during meiotic progression remains poorly understood. Polo-like kinases (PLKs) play widely conserved roles in orchestrating meiotic chromosome dynamics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
